Today, the HIV research community is one step closer to finding a cure. In a Canadian first, a team led by CTN+ researchers has reported a case of prolonged HIV remission following cancer treatment and a bone marrow transplant. The team, led by CTN+ National Co-director Dr. Sharon Walmsley (University Health Network) and CTN+ Researcher Dr. Mario Ostrowski (St. Michael’s Hospital, Unity Health, University of Toronto), reported their results in a late breaking abstract session at the 2026 Canadian Association for HIV Research (CAHR) conference in Winnipeg.
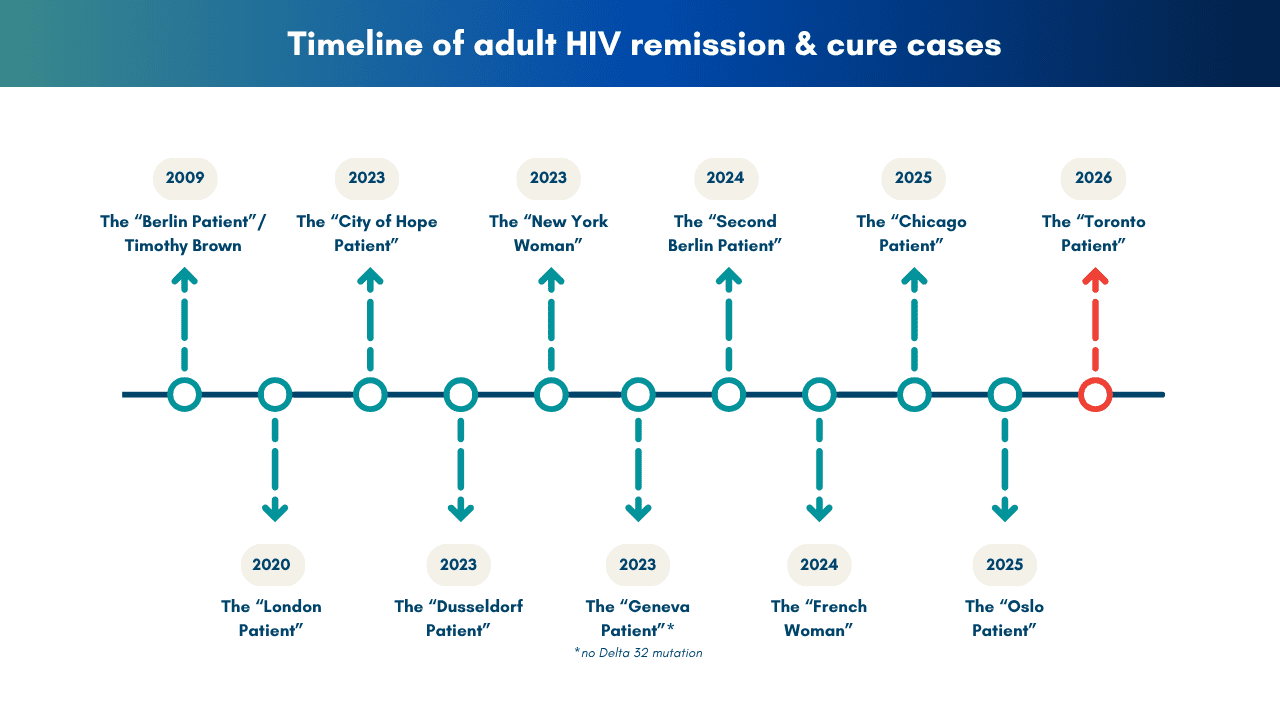
The patient, dubbed the Toronto Patient, who chose to remain anonymous, is the eleventh instance of someone whose HIV remains undetectable after undergoing cancer treatment followed by a bone marrow transplant. The patient stopped antiretroviral therapy (ART) in July 2025 and, as of April 2026, remains undetectable.
Dr. Walmsley, who has served as the Toronto Patients infectious disease doctor since 1999, says that the team is cautiously optimistic about viral suppression being maintained.
“He has been HIV-free for a little under year, so we are calling this ‘sustained remission,” she explained. “It is not exactly clear when we can call this a cure, but some people have suggested the three-year mark.”
The significance of the three-year horizon comes from the case of the Mississippi Baby, who was born with HIV and treated very early with antiretroviral therapy. Their HIV viral load remained undetectable for over two years before rebounding.
However, says Dr. Walmsley, the Mississippi Baby was cured using a very different approach, so the two cases are not exactly comparable.
Importantly, there is a chance that the Toronto Patient’s viral load could rebound. This was the case for the Chicago Patient, whose case was reported at CROI last year. His viral load rebounded but was quickly controlled by restarting ART. After two years on ART, he stopped treatment and has been in remission for nearly a year.
Extraordinary Odds
While this is the now the eleventh reported instance of HIV cure after bone marrow transplant, the chances of a case like this are astoundingly slim.
What these cases have in common is that they all received bone marrow donations from people who are naturally resistant to HIV. This resistance is due to a Delta‑32 genetic mutation in the CCR5 gene. This mutation disables CCR5, a key receptor on the surface of a person’s white blood cells that HIV uses to gain entry. Without a functioning CCR5 receptor to latch on to, HIV is effectively blocked from getting into the immune system.
“The theory is that this mutation evolved about 2,000 years ago as a protection from smallpox,” explained Dr. Walmsley. “Unfortunately, it’s only present in about 1% of people with European heritage.”
When a patient undergoes chemotherapy for cancer, the treatment almost entirely eradicates the person’s immune system. The patient’s immune system is then replaced using the donor’s bone marrow, where immune cells are created.
Aside from needing to find a donor with this rare mutation, a patient also needs to be compatible with the donor in order to receive the transplant with less chance of rejection. In the case of the Toronto Patient, a search of an international bone marrow registry resulted in a suitable match. The bone marrow transplant took place at Princess Margaret Hospital in Toronto.
“This case represents years of careful clinical work, close monitoring, and collaboration across specialties,” said hematologist Dr. Tommy Alfaro Moya in a UHN press release. “This case offers critical insight into how HIV can be eliminated from the body, informing safer approaches in the future.”
The potential impact of a donor with a CCR5 Delta‑32 mutation demonstrates how important it is to have CCR5 mutation information readily available in bone marrow donor registries.
“This calls for a worldwide registry of this mutation within the donor pool. We need a system whereby if someone anywhere in the world has HIV and needs bone marrow, we can quickly find out who is a match,” said Dr. Walmsley.
Where to From Here?
Patients undergoing a bone marrow transplant face a 10–20% mortality rate, meaning that a transplant from a donor with a CCR5 Delta‑32 mutation is clearly not a viable cure approach for the approximately 40 million people living with HIV worldwide.
“The transplant was performed to address the patient’s high-risk leukemia. A secondary, and extraordinary, outcome was finding a CCR5 mutated donor that allowed the patient to stop taking ART,” noted Dr. Alfaro Moya.
However, the case of the Toronto Patient also contributes valuable information to immunologists and HIV researchers who are searching for a cure.
“It’s important to study these cases intensively to get clues about the exact the mechanism of cure,” said Dr. Ostrowski, who led all of the research-related testing for this case at the University of Toronto.
Not every bone marrow transplant with this mutation has resulted in a successful cure, meaning that scientists have a lot to learn from each successful case.
According to Dr. Ostrowski, what is most remarkable about these cases is not how unique their circumstances are but the similarities between them. For example, the Toronto Patient and many of the other eleven cases, developed graft-versus-host disease after the transplant. “The job of immune cells is to attack foreign cells. Because you’re transplanting in a new immune system, it can see the body as foreign and attack it. This is called graft-versus-host disease,” he explained. In patients with HIV, a byproduct of this aggressive immune system is that the new immune cells it produces, called T cells, also attack the residual HIV-infected cells in the different viral reservoirs around the body.
“The virus can’t escape from these reservoirs because the new immune cells don’t have the CCR5 receptor for it to use, so it has nowhere to go,” said Dr. Ostrowski.
It’s research like this that helps to understand how these different circumstances interact and more clarity about the mechanics of the virus and immune system. This gives researchers important insights in developing new immunotherapeutic approaches, or refining older, previously unsuccessful strategies. For example, current research is attempting to use an mRNA vaccine to trigger a strong T-cell response against the virus, like occurs in graft-versus host disease.
“It’s cure cases like this that continue to fuel the idea that we could possibly cure patients without doing a bone marrow transplant,” said Dr. Ostrowski.
One step closer
“Some people ask, ‘Well, with well-tolerated, suppressive antiretroviral therapy now, HIV is controlled so well, why do we need a cure?’,” said Dr. Walmsley. “The reason is that we know that HIV contributes to other comorbidities and continues to have stigma associated with it.”
“HIV cure research is crucial because HIV is an inflammatory disease and causes chronic inflammation,” expanded Devan Nambiar, Community Co-lead of the CTN+ Cure & Immunotherapies Think Tank. “Even with an undetectable viral load, HIV can accelerate changes normally associated with aging, including non-AIDS-related cancers, heart disease, and declining bone health, as well as fragility and various other comorbidities.”
“These issues are compounded by stigma. As a person living with HIV for nearly 37 years, I am very encouraged by the research in Canada,” he added. “Personally, I would welcome life without daily HIV reminders, antiretroviral, and the stigma of HIV, and it will be a boost to mental, emotional, sexual, and physical health.
Specifically, people living with HIV are at an elevated risk of developing things like heart disease and cancer compared to people not living with HIV, including 15 times more likely to develop Burkitt lymphoma, the type of cancer the Toronto Man first presented with in 1999.
After beating the first round of cancer, he then developed leukemia in 2021 which led to the bone marrow transplant, making him the only HIV cure case who survived two types of cancer.
The Toronto Patient’s extraordinary journey exemplifies the importance of research to understand, treat, and prevent comorbidities and age-related diseases in people living with HIV. As a participant in CHANGE HIV (CTN 314), the first geriatric HIV cohort in Canada, he has now contributed vital information to multiple aspects of HIV research.
But his key contribution as one of the very few cure cases is to remind the world that HIV continues to affect the lives of millions worldwide and to push the conversation and field forward into the future.