CTN 222: Canadian co-infection cohort
A prospective clinical cohort of HIV and hepatitis C virus co-infected patients
About The Study
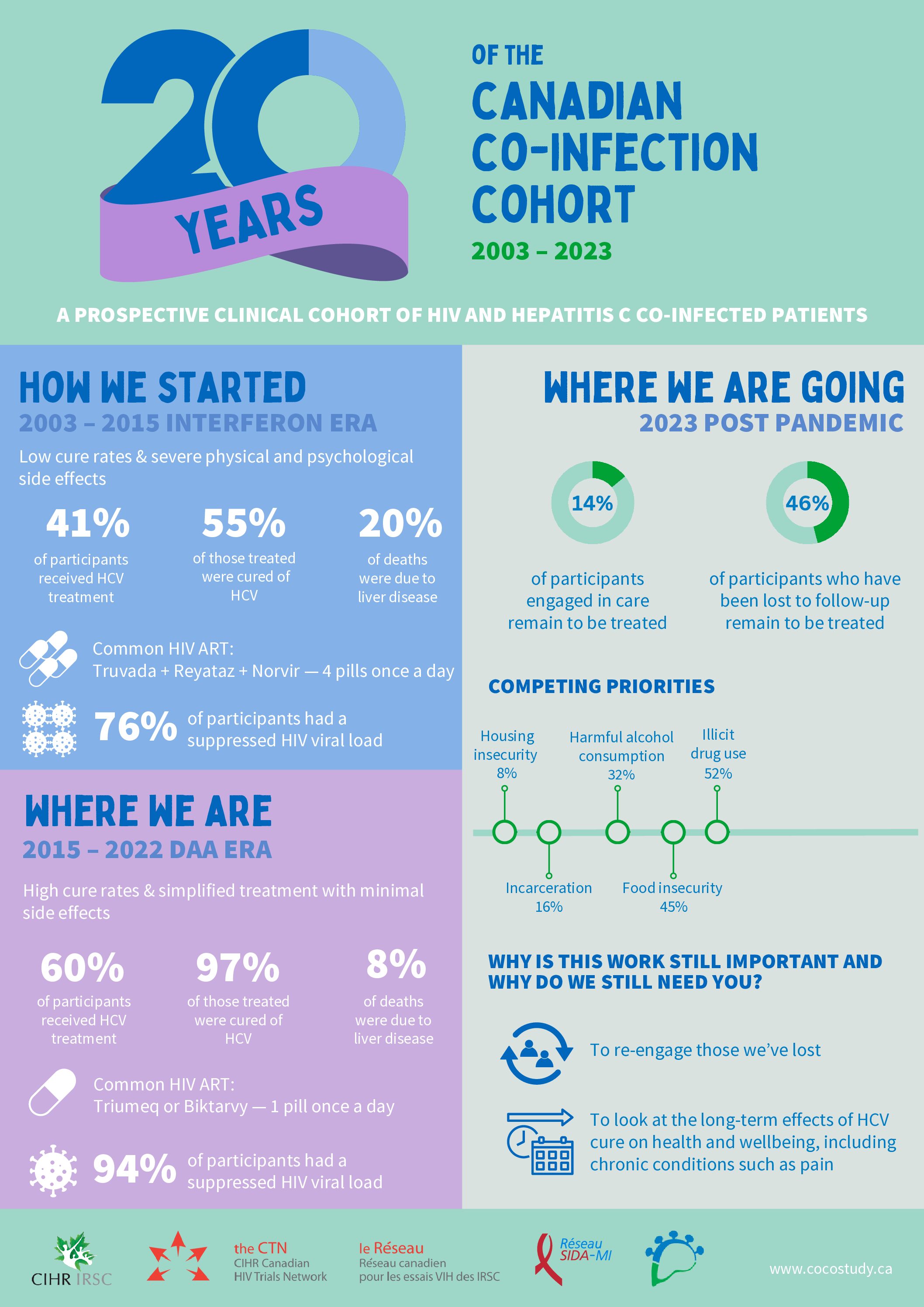
Initiated in Montreal in 2002 and funded by CIHR in 2005, the Canadian Co-infection Cohort Study received support from the CTN starting in 2007. This national cohort study examines the effect of antiretroviral therapy (ART) on liver disease progression in people living with both HIV and hepatitis C virus (HCV). CTN 222 was the first observational cohort study approved by the CTN and included 950 participants from across 16 sites. With renewed funding from CIHR in 2009 and again in 2014, CTN 222 has expanded to include over 1600 people living with HCV and HIV.
Learn MoreBackground
HCV is a virus that attacks the liver, causing liver disease (hepatitis). Because HCV can be contracted through the same routes as HIV (blood-to-blood contact, sexual contact, etc.) over 30% of people living with HIV worldwide also have HCV. Due to the success of antiretroviral therapies (ART), complications from comorbidities such as HCV have replaced AIDS-related infections as the primary health concern among people living with HIV.
End-stage liver disease (ESLD) due to HCV is one of the leading causes of illness and death in HIV/HCV+ people. The natural course of HCV is accelerated in people living with HIV. Initially, the ability of ART to restore the immune system was expected to improve liver health in this population. This was not the case, however, and liver disease continues to be a significant problem. In response, CTN 222 was formed to document and understand the social and biological factors that contribute to the interaction between these two viruses and their treatment and management.
Study Approach
CTN 222 is a long-term observational cohort study and includes a diverse group of people including men who have sex with men (MSM), current and ex-injection drug users, Indigenous people, and women. All eligible individuals who visit a participating clinic are invited to participate in the study. Those who initially decline the invitation are eligible to enrol in the future.
After giving informed consent and undergoing an initial evaluation, CTN 222 participants return approximately every 6 months, either as a part of clinical care or for study purposes alone. Clinical, sociodemographic, behavioural, medical, and treatment information is collected. Treatment and diagnosis history is also collected from past medical visits.
Results
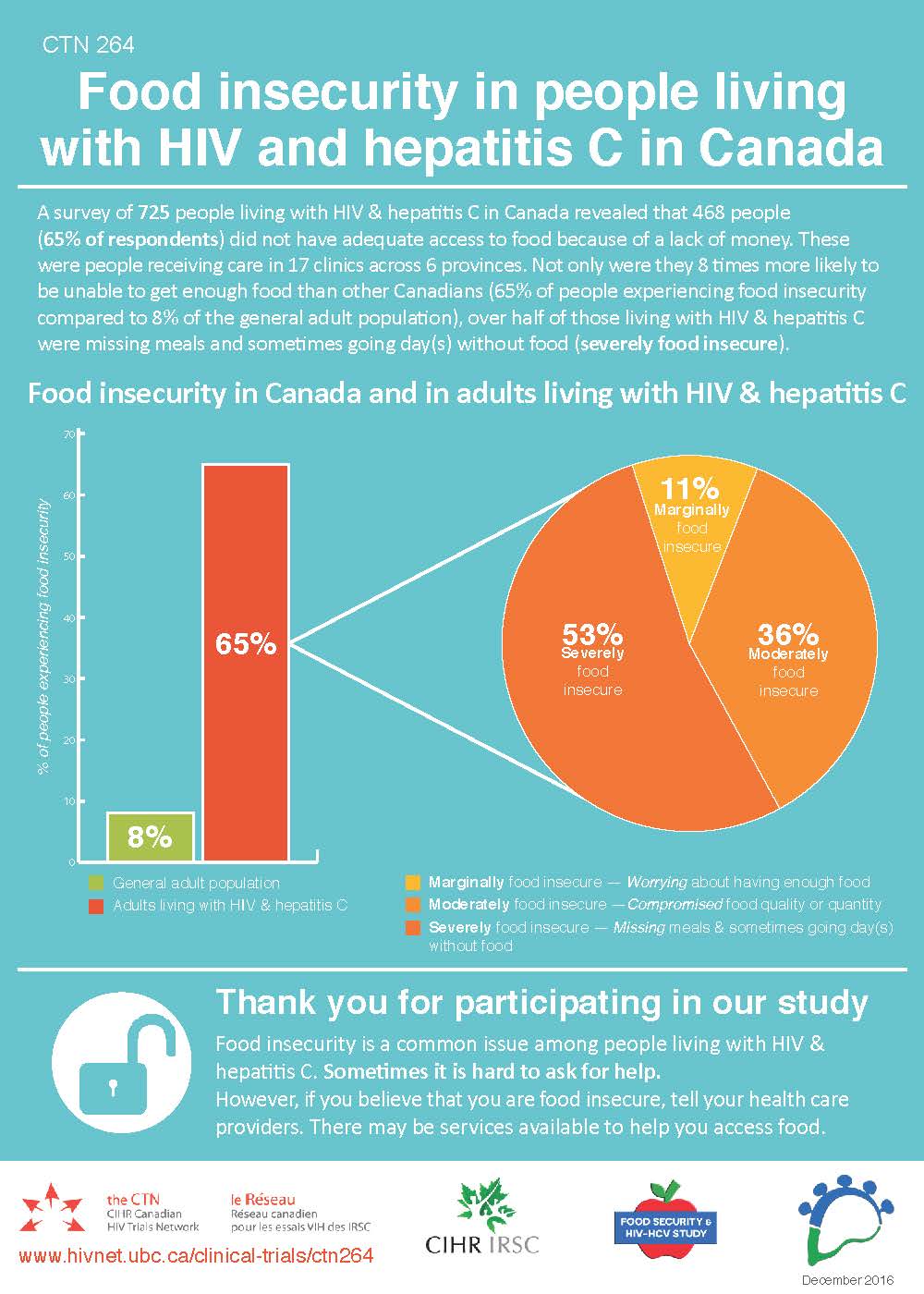
Since 2010, the CTN 222 team has published over 30 journal articles. This body of work is expansive, stretching across different areas of this complex area; this includes general cohort information, the effect of different treatment regimens, the impact of immunological factors, genetic variations, and lifestyle factors, and the interaction between participant sub-groups and liver-related health. One such characteristic is the effect of food insecurity on HIV-HCV outcomes which has spawned a CTN 222 sub-study, CTN 264.
Additional Information
You can contact the CTN 222 team via email at cviscoco@gmail.com and on X at our handle @cocostudy. We also encourage you to watch this presentation to learn more about the history of the Canadian Co-infection Cohort.
Background
HCV is a virus that attacks the liver, causing liver disease (hepatitis). Because HCV can be contracted through the same routes as HIV (blood-to-blood contact, sexual contact, etc.) over 30% of people living with HIV worldwide also have HCV. Due to the success of antiretroviral therapies (ART), complications from comorbidities such as HCV have replaced AIDS-related infections as the primary health concern among people living with HIV.
End-stage liver disease (ESLD) due to HCV is one of the leading causes of illness and death in HIV/HCV+ people. The natural course of HCV is accelerated in people living with HIV. Initially, the ability of ART to restore the immune system was expected to improve liver health in this population. This was not the case, however, and liver disease continues to be a significant problem. In response, CTN 222 was formed to document and understand the social and biological factors that contribute to the interaction between these two viruses and their treatment and management.
Study Approach
CTN 222 is a long-term observational cohort study and includes a diverse group of people including men who have sex with men (MSM), current and ex-injection drug users, Indigenous people, and women. All eligible individuals who visit a participating clinic are invited to participate in the study. Those who initially decline the invitation are eligible to enrol in the future.
After giving informed consent and undergoing an initial evaluation, CTN 222 participants return approximately every 6 months, either as a part of clinical care or for study purposes alone. Clinical, sociodemographic, behavioural, medical, and treatment information is collected. Treatment and diagnosis history is also collected from past medical visits.
Knowledge Mobilization
Impact
Publications from this study have been cited and used to inform policy and care decisions across the world — below are a few key examples of this reach and impact.
Guidelines for the screening, care and treatment of persons with hepatitis C infection
World Health Organization
Guidelines for the use of antiretroviral agents in HIV-1-infected adults and adolescents
Centre for Disease Control (CDC)
Oral Presentations
You can download the full list of oral presentations from 2023 to 2005 here.
Live Video Presentations
Charlotte Lanièce Delaunay – CROI Conference – March 2021
Gayatri Marathe – Infectious Diseases and Immunity in Global Health (IDIGH) Lunch Seminar – December 2021
Infographics
You can download the infographics related to the study here.
Eligibility Requirements
- Aged 16 years or older (or as provincial guidelines)
- Documented HIV seropositive infection
- Documented HCV infection or evidence of exposure
- Able to provide informed consent
Purpose of the study:
- To improve the health of people living with HIV-HCV
- To improve access to care
Participation in the study takes 45 minutes and involves:
- Blood sample (standard of care)
- Fibroscan (health of your liver)
- A brief questionnaire
You will be compensated for your time/travel $25/visit (follow-up every 6 months)
The Team
You can also find the list of trainees (active and past) who participated in the study by clicking on the link here.
info@ctnplus.caParticipating Sites
Current Sites
Southern Alberta Clinic
- Dr. John Gill
- Calgary, AB
- Contact Person
- contact@email.com
- 403-955-6309
SHARE University of Saskatchewan
- Dr. Cara Spence
- Saskatoon, SK
- Contact Person
- contact@email.com
- 306-717-3679
Regina General Hospital
- Dr. Alexander Wong
- Regina, SK
- Contact Person
- contact@email.com
- 306-766-3915
West Side Community Clinic
- Dr. Larissa Kiesman
- Saskatoon, SK
- Contact Person
- contact@email.com
- 306-664-4310
Oak Tree Clinic
- Dr. Katherine Plewes
- Vancouver, BC
- Contact Person
- contact@email.com
- 604-875-2212
Vancouver Infectious Diseases Centre
- Dr. Brian Conway
- Vancouver, BC
- Contact Person
- contact@email.com
- 604-642-6429
BC Centre for Excellence in HIV/AIDS, St. Paul’s Hospital
- Dr. Mark Hull
- Vancouver, BC
- Contact Person
- contact@email.com
- 604-506-8477
Royal Victoria Hospital
- Dr. Joseph Cox
- Montreal, QC
- Contact Person
- contact@email.com
- 514-843-2090
McMaster University Medical Centre
- Dr. Shariq Haider
- Hamilton, ON
- Contact Person
- contact@email.com
- 905-521-2100
Health Sciences North (The HAVEN Program)
- Tammy Bourque
- Sudbury, ON
- Contact Person
- contact@email.com
- 705-523-7077
University Health Network – Toronto General Hospital
- Dr. Sharon Walmsley
- Toronto, ON
- Contact Person
- contact@email.com
- 416-340-4800
Windsor Regional Hospital
- Dr. Corinna Mae Quan
- Windsor, ON
- Contact Person
- contact@email.com
- 519-254-6115
The Ottawa Hospital (General Campus)
- Dr. Curtis Cooper
- Ottawa, ON
- Contact Person
- contact@email.com
- 613-737-8899
McGill University Health Centre (Glen Site)
- Dr. Marina Klein
- Montreal, QC
- Contact Person
- contact@email.com
- 514-843-2090
CHUM – Hôtel-Dieu
- Dr. Valérie Martel-Laferrière
- Montreal, QC
- Contact Person
- contact@email.com
- 514-890-9000
CHU de Québec-Université Laval
- Dr. Marie-Louise Vachon
- Québec City, QC
- Contact Person
- contact@email.com
- 418-525-4444 ex. 47778
Queen Elizabeth II Health Sciences Centre
- Dr. Lisa Barrett
- Halifax, NS
- Contact Person
- contact@email.com
- 902-470-8888
- Site Investigator
-
12345 Street Name,
City Name, BC V3M 9J3
- Contact Person
- contact@email.com
- 1 (604) 555-9999
Past sites
Vancouver Aboriginal Health Society
- Dr. Mark Hull
- Vancouver, BC
- Contact Person
- contact@email.com
- 604-254-9949
Clinique Médicale du Quartier Latin
- Dr. Pierre Coté
- Montreal, QC
- Contact Person
- contact@email.com
- 514-285-5567
- Site Investigator
-
12345 Street Name,
City Name, BC V3M 9J3
- Contact Person
- contact@email.com
- 1 (604) 555-9999
Resources
Available Services
HIV-HCV Co-infection Slide Kit
Evolution and Revolution: Current Issues in HIV and HCV Co-infection
DownloadCommunity Partnerships
The Canadian Co-infection Cohort is proud to partner with both local and national community organizations. Our collaborators include CATIE, AIDS Community Care Montreal (ACCM), and the “Coalition des organismes communautaires Québécois de lutte contre le sida” (COCQ-SIDA).
Established in 1990, CATIE’s mandate has expanded to providing both treatment and prevention information on HIV and hepatitis C to frontline service providers across Canada. CATIE strengthens Canada’s response to HIV and hepatitis C by bridging research and practice. They connect healthcare and community-based service providers with the latest science and promote good practices for prevention and treatment programs.
Established in 1987, ACCM is a volunteer-driven community organization that provides support services and treatment information to people living with HIV/AIDS and/or hepatitis C. The voices of their members are central to ACCM’s guidance, and they work in collaboration with their many communities to build a compassionate and caring response to HIV and hepatitis C.
Established in 1990, the COCQ-SIDA’s mission is to bring together Quebec community organizations involved in the fight against HIV/AIDS and exercise its leadership in order to encourage, support, consolidate, and promote autonomous community action in the fight against HIV/AIDS in the Quebec territory.
International Collaborations
The Canadian Co-infection Cohort is proud to be included in two international cohorts: InCHEHC and Hep-CAUSAL.
The Burnet Institute’s International Collaboration on Hepatitis C Elimination in HIV Cohorts (InCHECH) pools data from 15 observational cohorts from Australia, France, Spain, Switzerland, the Netherlands, Georgia, the United States, and Canada. It is the largest international cohort of people living with HIV with well-characterized data on HCV co-infection.
Pooling data from approximately 75,000 participants, the aim of this sizeable cohort is to measure and identify correlates of rare outcomes that cannot be analyzed within individual cohorts or countries, such as HCV reinfection after successful treatment, treatment failure, and liver-related mortality.
InCHEHC also allows for the interim assessment of the WHO elimination targets in 2020-2022, facilitating refinement of policy and practice within the target period.
Harvard’s School of Public Health’s Hep-CAUSAL is a consortium of cohorts of people who are co-infected with HIV and HCV in Europe and the Americas. By combining observational data and causal inference methods, Hep-CAUSAL estimates the long-term risk of HCV reinfection, hepatic disease, and extra-hepatic disease for all co-infected people. The findings are set to inform post-cure management guidelines and how to maximize the benefits of DAA treatment in the long term.